This typically brings the optical densities back within the straight-line portion of the film’s sensitometric curve to best visualize the anatomic area of interest. In general, for repeat radiographs necessitated by density errors, the mAs is adjusted by a factor of 2 therefore a minimum change involves doubling or halving the mAs. The radiographer must decide how much of a change in mAs is needed to correct for the density error. When the radiograph is deemed unacceptable, this means the optical densities lie outside the film’s sensitometric curve’s straight-line portion, and may need to be repeated. When using a film-screen IR, radiographers need to assess the level of density produced on the processed image and determine whether the density is sufficient to visualize the anatomic area of interest. The film characteristic, speed, and chemical processing determine the amount of optical density produced on the image for a given mAs. This relationship between radiation exposure intensity and density is discussed in more detail in Chapter 9. When a film image is too light (insufficient density), a greater increase in mAs may be needed to correct the density, or the mAs may need to be decreased to correct a film image that has excessive density. (From Fauber TL: Radiographic imaging and exposure, ed 3, St Louis, 2009, Mosby.) C, Increase in density when the mAs is doubled. B, Decreased in density when the mAs is decreased by half. the structure of the material, XRD can be used for qualitative and quantitative phase identification.FIGURE 10-2 mAs and Radiographic Density.Ĭhanges in mAs have a direct effect on density. Since the distances between the atomic planes are dependent on the size and distribution of atoms – i.e. It is the scattering from these periodic arrays that leads to the diffraction effect, and there is a simple relationship (Bragg's law) between the scattering angle, the wavelength of the radiation and the spacings between the planes of atoms. All substances are built up of individual atoms and nearly all substances have some degree of order of periodicity in the arrangement of these atoms. Where the scattered wavelengths interfere with one another diffraction of X-rays occurs. XRF is a technique which utilizes the diffracting power of a single crystal, or the proportional characteristics of a photon detector, to separate the polychromatic beam of radiation from the sample into separate wavelengths, thus allowing qualitative and quantitative elemental measurements to be made.Ī beam of monochromatic radiation may also be scattered when X-ray photons collide with atomic electrons. Thus when a sample is made up of many different types of atoms, each atom will produce a series of wavelengths, and all of the contributions add up to become the total X-ray emission from the sample.  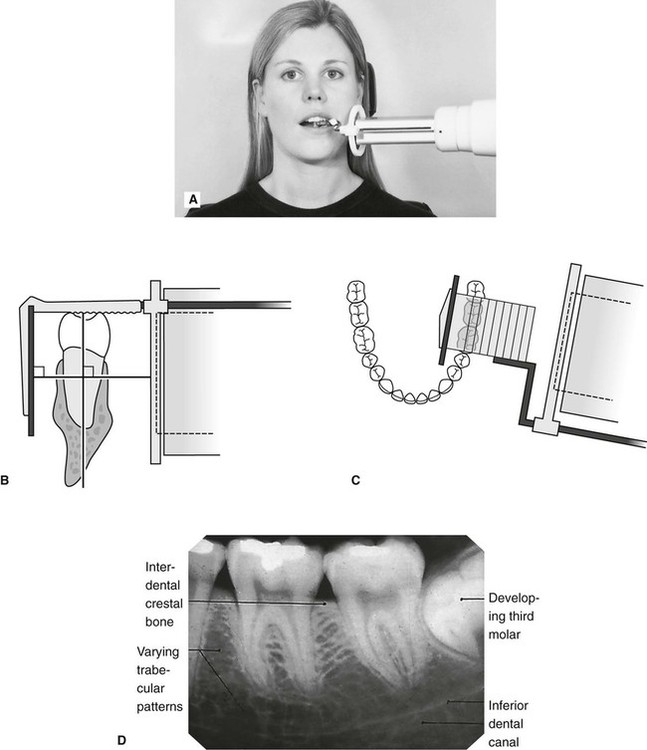
The X-ray emission wavelengths are characteristic of the atom in question and there is a simple relationship (Moseley's law) between the wavelength of the emission line and the atomic number of the atom. Each of these transitions is accompanied by the emission of an X-ray photon having an energy equal to the energy difference between the two states. One of the processes by which the atom regains stability is by transference of electrons from outer to inner electron shells. When a sample of material is bombarded with energetic radiation (X-rays, γ-rays, electrons, protons, etc.) vacancies may arise from the removal of inner orbital electrons. While the broad definition of X-ray methods covers many techniques based on the scatter, emission and absorption properties of X-radiation, the two most common are X-ray fluorescence (XRF) spectrometry and X-ray powder diffractometry (XRD). The use of X-ray methods in the field of materials analysis is now entering its seventh decade.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
